Efficacy and Safety of a Novel Gene Therapy (URO-902; pVAX/hSlo) in Female Patients With Overactive Bladder and Urge Urinary Incontinence: Results From a Phase 2a Trial
Hanh Badger, PharmD1, Kenneth M. Peters, MD2, Ekene A. Enemchukwu, MD, MPH3, Susan Kalota, MD4, Kaiser Robertson, MD5, Heather Greene, MPH1, Salim Mujais, MD1, Cornelia Haag-Molkenteller, MD, PhD1.
1Urovant Sciences, Irvine, CA, USA, 2Oakland University William Beaumont School of Medicine, Rochester, MI, USA, 3Stanford University School of Medicine, Stanford, CA, USA, 4Urologic Associates of Southern Arizona, Tucson, AZ, USA, 5Chesapeake Urology, Hanover, MD, USA.
Introduction: URO-902 is an investigational gene therapy for overactive bladder (OAB) and consists of a plasmid vector that expresses the α subunit of the large-conductance Ca2+-activated K+ channel in the detrusor to reduce bladder hypercontractility. We report interim results from a phase 2a trial assessing the safety and efficacy of URO902 in OAB. Materials & Methods: This was a prespecified, 12-week interim analysis of a 48-week multicenter, randomized, double-blind, placebo-controlled, dose-escalation study (NCT04211831). Women aged 40‒79 years with OAB and urge urinary incontinence (UUI) who were not adequately managed with oral OAB medications were randomly assigned to receive single-dose URO-902 24 mg, URO-902 48 mg, or placebo administered by intradetrusor injection via cystoscopy under local anesthesia. Safety was assessed by adverse events (AEs) and postvoid residual (PVR) urine. Exploratory endpoints included change from baseline (CFB) to week 12 in mean daily micturitions, urgency episodes, UUI episodes, and quality of life (QoL) measures. Results: Of the 80 patients randomized, 68 completed week 12, and 74 were included in the intent-to-treat population. Mean (SD) age was 64.7 (7.1) years, and 13.5% had prior treatment with onabotulinumtoxinA. At week 12, URO-902 24 and 48 mg were associated with clinically relevant improvement vs placebo in mean daily micturitions (LS mean CFB, ‒2.5 and ‒3.0 vs ‒1.0, respectively), urgency episodes (‒2.4 and ‒3.4 vs ‒1.2), UUI episodes (‒2.4 and ‒2.6 vs ‒2.3), OAB questionnaire symptom bother score (‒24.1 and ‒25.3 vs ‒11.2), and proportion of patient global impression of change responders (40.9% and 57.7% vs 30.8%) (Table). Treatment-emergent AEs occurred in 45.5% of patients receiving URO-902 24 mg, 46.2% receiving 48 mg, and 50.0% receiving placebo. The most commonly occurring AE (24 mg/48 mg/placebo) was urinary tract infection (0%/15.4%/3.8%). One patient (48-mg arm) had asymptomatic elevated PVR urine volume at week 2 (resolved spontaneously, did not require catheterization). Conclusions: In this phase 2a trial of women with OAB and UUI, a single dose of URO-902 24 or 48 mg was associated with clinically relevant improvement in efficacy and QoL endpoints and was safe and well tolerated. 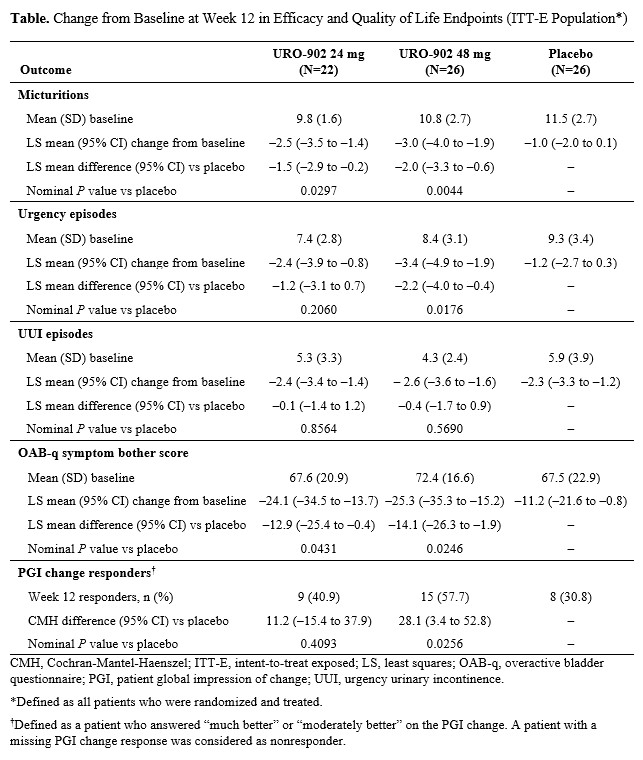
Back to 2022 Abstracts


