Back to 2017 Program
Urothelial genes modulating micturition behavior induced by a repetitive lipopolysaccharide (LPS) exposure in an ovariectomized (OVX) mouse model
Marian Acevedo, MD1, Judy Yeh, MD1, Lery Alvarez-Lugo, MS1, Ming Lu, MD1, Nitin Sukumar, MS1, Warren Hill, PhD2, Toby C. Chai, MD1.
1Yale University, New Haven, CT, USA, 2Harvard Medical School, Boston, MA, USA.
BACKGROUND: Menopause increases risk of recurrent UTI (rUTI) which manifests with chronic lower urinary tract symptoms (LUTS). While treatment focuses on eradicating pathogens (e.g. antibiotics), we lack an understanding of host responses in this scenario which can lead to bladder functional changes, including refractory LUTS. In this study, we sought to measure host responses, including voiding behavior, urothelial gene expression, and bladder morphology. We identified urothelial transcripts influenced by OVX state that regulated voiding behavior after repetitive LPS intravesical exposure.
METHODS: Female C57BL6 mice underwent sham (n=10) or OVX (n=10) surgery. Micturition behavior was measured using voiding spot assay (VSA) performed pre-surgery, 4 weeks post-surgery (but prior to LPS exposure) and after each of three consecutive days of intravesical inoculation of LPS. At end of experiment, animals were euthanized and bladders harvested for Gomori trichome staining. A separate experiment, following same LPS exposure protocol, was performed in 18 mice (sham=9, OVX =9) for microarray analysis of urothelial gene expression (pure urothelial sheet dissections) before LPS, 1d and 3d after LPS exposure, using Affymetrix gene chip for entire mouse transcriptome.
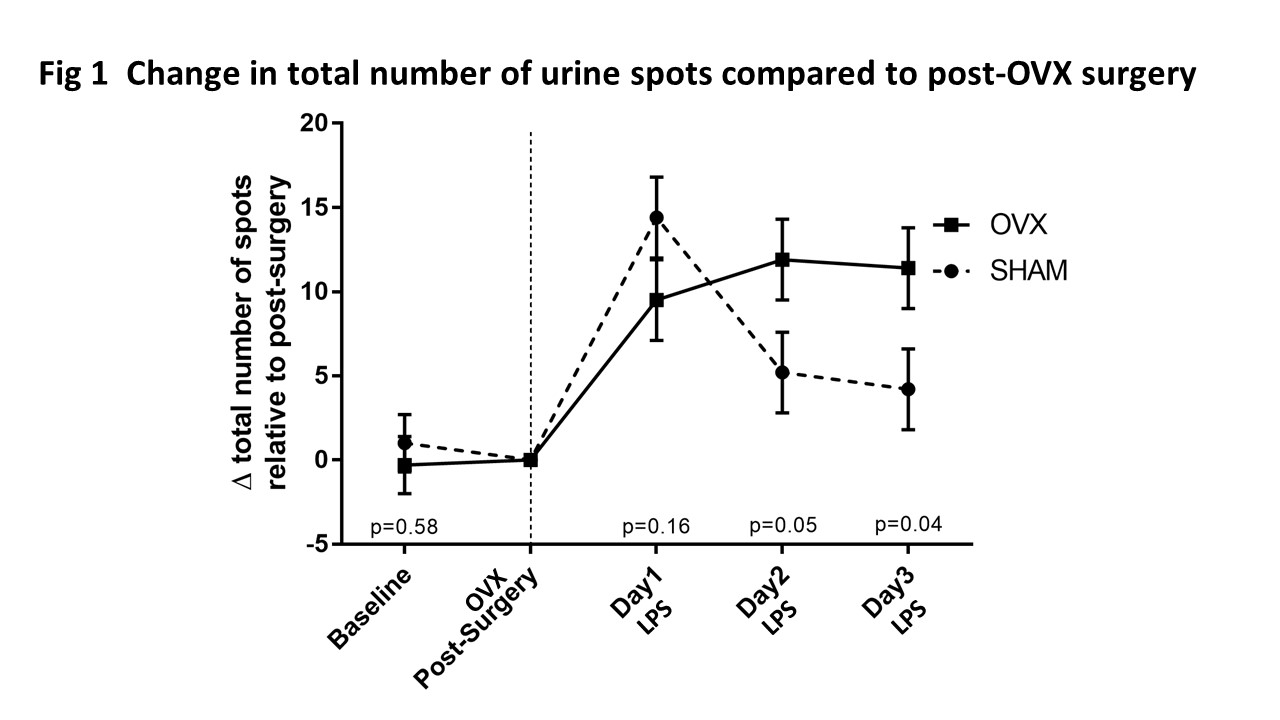
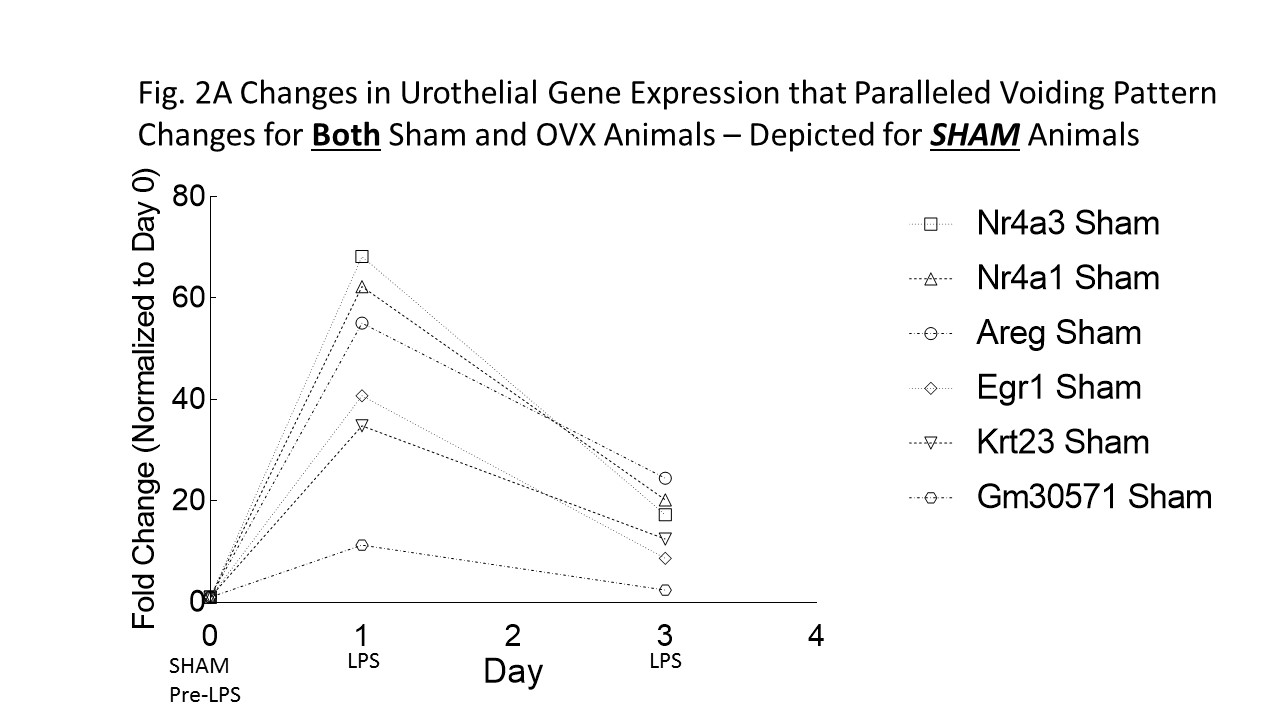
RESULTS: In Fig 1, OVX and sham animals exhibited overactive voiding behavior on day 1. However, OVX and sham animals diverged on days 2 and 3 of LPS treatment with the OVX mice persisting with an overactive voiding phenotype. Gomori trichome staining showed that OVX mice had flattened rugae which was not seen in sham mice. Analysis of microarray data focused on pattern of gene expression changes (cutoff >+10x or <-10x) that mimicked voiding pattern changes for both sham and OVX animals. Only 6 transcripts, out of the entire transcriptome surveyed, were identified using this focused approach: Nr4a3 (nuclear receptor 4a3), Nr4a1 (nerve growth factor IB), Areg (amphoregulin), Egr1 (early growth response 1), Krt23 (keratin type 1 cytoskeletal 23), and Gm30571 (unknown protein). These 6 transcripts followed the pattern seen in Fig. 1 (voiding behavior) for both sham (Fig. 2A) and OVX animals (Fig. 2B). For example, urothelial transcript Nr4a3 in sham animals (Fig. 2A, open box) went up (65x) 1 day after LPS, but came down (20x) 3 days after LPS. However, this same transcript in OVX animals (Fig. 2B, closed box) went up (38x) 1 day after LPS and persisted (38x) 3 days after LPS. Thus changes in Nr4a3 mimicked the VSA data shown in Fig. 1.
CONCLUSIONS: Targeted microarray analysis revealed only 6 urothelial gene expression changes that uniquely paralleled voiding changes. These genes involve inflammation, epithelial growth/repair and cytokines. Treating LUTS secondary to inflammation/UTI might target these host response urothelial genes.
Back to 2017 Program
|
|
|
|
|






