| NEAUA Main Site | | | Past & Future Meetings |
|

|
Leveraging Big Data to Study Bladder Cancer Care Florian R. Schroeck, MD, MS1, Brenda Sirovich, MD, MS2, John D. Seigne, MBBS3, Douglas J. Robertson, MD, MPH2, Philip P. Goodney, MD, MS2. 1White River Junction VA Medical Center, White River Junction, VT, and Section of Urology and Norris Cotton Cancer Center, Dartmouth Hitchcock Medical Center, Lebanon, NH, USA, 2White River Junction VA Medical Center, White River Junction, VT, USA, 3Section of Urology and Norris Cotton Cancer Center, Dartmouth Hitchcock Medical Center, Lebanon, NH, USA.
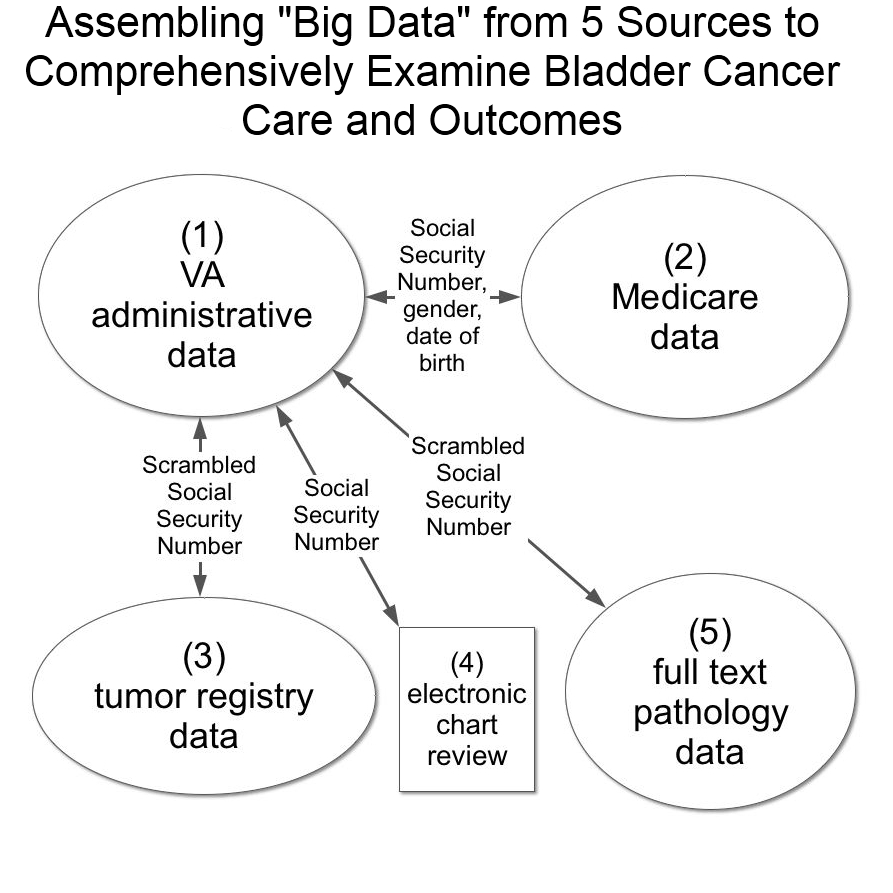
BACKGROUND: Despite the high prevalence of bladder cancer, research on optimal bladder cancer care is limited. One way to advance observational research on care is to use linked data from multiple sources. Such “big data” research can provide real-world details of care and outcomes across a large number of patients. We assembled such data including (1) administrative data from the Department of Veterans Affairs (VA), (2) Medicare claims, (3) data abstracted by tumor registrars, (4) data abstracted via chart review from the national electronic health record, and (5) full text pathology reports. Based on these combined data, we validated the use of administrative data to identify newly diagnosed bladder cancer patients who received care in the VA. Back to 2017 Program |
|
Photos courtesty of Old Port of Montréal © Tourisme Montréal, Stéphan Poulin.